Uses:
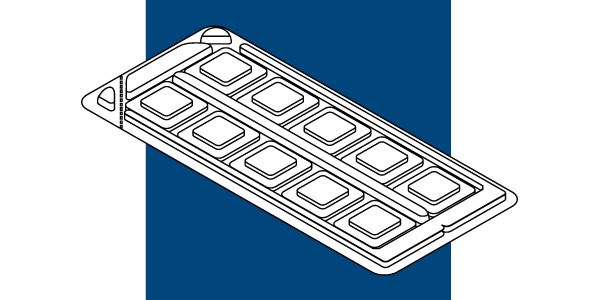
IQ Ultra™ is the comfortable and reliable Patch Test Unit choice for the aid of diagnosis of contact allergy. The IQ Ultra™ suitable for preloading features the advanced IQ Chambers mounted on medical grade carrier tape.
Product features:
Adhesive chamber rim
The medical grade acrylic adhesive helps prevent hapten leakage and enhances the adhesion.
Foam frame for high comfort
The chambers are made of soft polyethylene foam for patient comfort.
Integrated filter paper
The integrated filter paper facilitates handling of liquid haptens.
Preloadable
Preloading increases efficiency as multiple tests can be prepared in advance. By using the Application Device™, loading of the test units is made efficient and time saving.
Aluminum free
IQ Ultra™ does not have uncomfortable metal chambers that might react chemically to haptens.
Medical grade material used
IQ Ultra™ use medical grade acrylic adhesives and non-woven carrier tape.
IQ Ultra™:
Quantity: 100 Patch Test Units
Size (mm) 52 x 118
IQ Chambers / Unit: 10 pcs
Rec. dose / IQ Chamber: 25 μl



